Blogger: Summer scientist* Espen Verpe
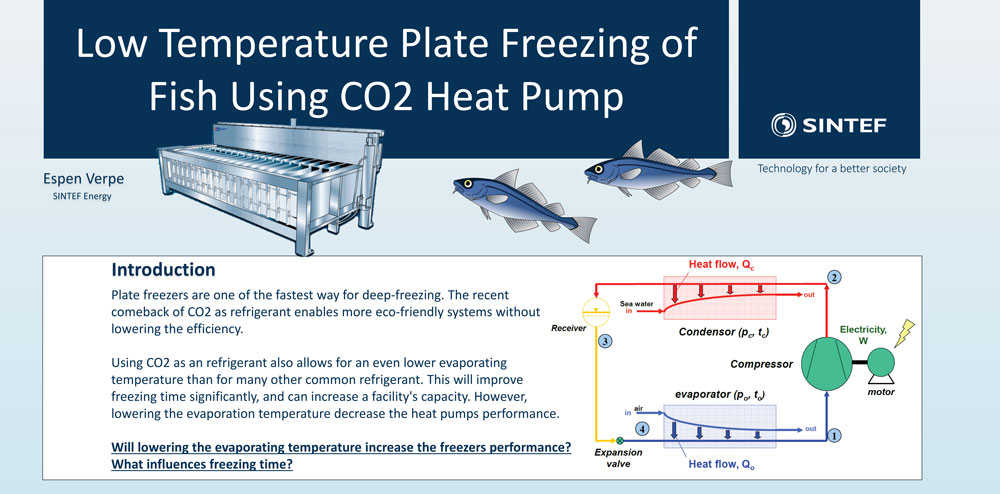
Freezing is one of the main technologies for food preservation and a significant number of products in a supermarket are sold in frozen state. Freezing is quite energy demanding since a refrigeration system must exchange and remove all the energy necessary to transform the water in the product to ice. In fact, 15% of the global electricity consumption is related to chilling and freezing.
System efficiency is not high
Conventional refrigeration systems are normally operated at temperatures around -30°C, because of the thermal limitations of the refrigerant. A special characteristic of the refrigerant R744, which is carbon dioxide, is that it could be used at temperatures down to -50°C, however the system efficiency is not high.
On the other hand, the freezing process will be faster at such low temperatures, since the temperature gradient between product and refrigerant is much higher.
The question to be answered is: What is more efficient? Freezing fast with low refrigeration efficiency or freezing slow with high refrigeration efficiency.

I was looking into this question during my stay at SINTEF Energy Research. My results clearly indicates that the freezing time reduction and the decrease in refrigeration efficiency are equal.
For a given plate freezing system this means that is possible to freeze more product with only a small increase in energy consumption for a given amount of product. The estimated capacity increase can be as much as 54 %, depending on what product is frozen, when comparing to freezing with a temperature of -30°C.
Two-dimensional freezing model
In order to gain this result, it was necessary to develop a two-dimensional freezing model that could estimate the freezing time at different refrigeration temperatures. The simulation results than had to be implemented into a heat pump model, comparing conventional refrigeration systems, using ammonia, with carbon dioxide refrigeration.
The promising results will be investigated further and it is planned to investigate and document the estimated capacity increase through field tests.
HighEFF
This work was a part of the Research Centre “HighEFF: Centre for an Energy Efficient and Competitive Industry for theFuture” one of Norway’s Centres for Environment-friendly Energy Research (FME) co-funded by the Research Council of Norway and Industry. The host institution is SINTEF Energy Research.
HighEFF will help ensure that Norway has the world’s greenest industries.
- 20% to 30% reduction in energy consumption
- 10% reduction of greenhouse gas emissions
- Increased value creation in Norway
- Energy efficient processes and components
- Competitive Norwegian industry
*Summerscientists at SINTEF Energy Research
For the 11th year in a row, SINTEF Energy Research has offered summer jobs to a number of students. This year we had as many as 32 students, all of whom were given the opportunity to be a research scientist for a summer. We like to call them “summer researchers”. In 2017 we received about 250 applications for jobs as summer researchers, so there was tough competition this year too.
- Summer jobs involve carrying out specific research tasks that are integral to our projects. Students are under the continual supervision of experienced SINTEF researchers.
- Summer researchers are integrated into the research teams.
- They give a joint presentation at a closing seminar in front of relevant SINTEF research scientists.
- They have the opportunity to carry out project-related and Master’s degree assignments as extensions of their summer job (this applies only to those jobs where this option is specified).
- Summer researchers must be proficient in Norwegian and English, both written and spoken.


0 comments on “Fast and efficient: Low temperature freezing at -50°C”