Guest bloggers: Dr. Michael Hentscher, GFZ German Research Centre for Geosciences and Dr. Svenja Waldmann, TNO
The researchers take part in the IMPACTS project: IMPACTS is a collaborative project co-funded by the European Commission under the 7th Framework Programme. IMPACTS is run by SINTEF Energy Research. Find out more about IMPACTS.
The first question we have to answer: What does geochemical influence during storage of impure CO2 mean? The geochemical influence describes chemical reactions of the gas injected in the storage formation and the dissolution of the reaction products into the water phase. This results in increasing water acidity, which furher enhances mineral dissolution reactions? Consequently the formation water chemistry changes over time and additional minerals may precipitate.
The mineral dissolution and precipitation reactions are important processes, which may have an influence on the storage conditions, i.e. changes in porosity and permeability of the reservoir rock. The dissolution of minerals during CO2-discharge is a favored process because it increases the pore volume and therefore it may have a positive influence on the permeability. Due to these mineral dissolution reactions, cations like Ca, Mg, Fe, and Mn may be concentrated in the water and can precipitate in form of carbonates. Carbonate precipitation is the safest option to store CO2 on the long-term.
The geochemical influence of impurities, like SOx, NOx, O2, and H2 in the CO2 stream on water-rock interactions is questionable. Possible processes are the acceleration of mineral dissolution as well the decrease of permeability due to mineral precipitation. Especially, impurities that dissolve and react with the formation water, like CO2 to carbonic acids. Such Impurities are for example SO2 and NO2, which react with water and form acids that are stronger than carbonic acids.
Experiments are useful tools to investigate the influence of impurities under controlled reservoir conditions. In the IMPACTS project, we use batch reactors that are put under high temperature and pressure conditions covering the reservoir situation.
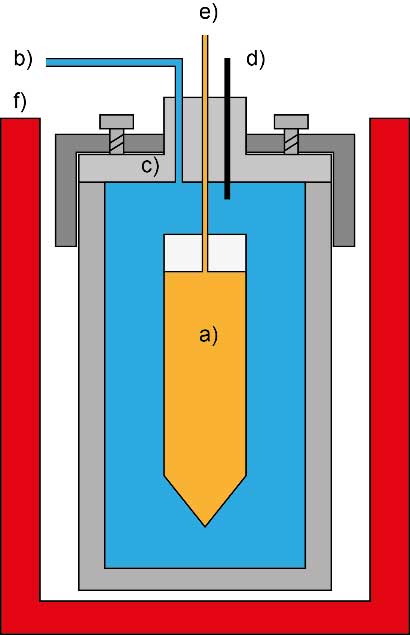
The figure shows a schematic configuration of a batch reactor with a deformable reaction cell fitted in a pressure bomb and encased by pressurized water. The enclosed reaction cell is loaded with the mineral or rock sample, a synthetic brine and the investigated gas mixture. Temperature and pressure of the storage formation is adjusted by heating of the batch reactor and by pressurization of the reaction cell surrounded by water. The water pressure compresses the reaction cell so that the same pressure is attained in the cell.
The experimental design allows fluid sampling from the reaction cell during the experimental run-time. However, the number of samples is restricted to the volume of the reaction cell, because the cell should not be more compressed than to 40% of its original volume.
The analysis of the water samples, the comparison the recovered mineral or rock sample before and after reaction enable us to draw conclusions about the geochemical impacts of impurities. It should be kept in mind that only short time reactions can be analyzed, which are of such character that they can be attained during the experiment. In addition, these experiments have to be repeated with and without impurities to distinguish the related effects from the impurities on the geochemical impact of CO2.

0 comments on “How we study the effects of CO2 impurities in the Lab?”