While much of the public focus rests on energy generation, the metallurgical industries—specifically those producing silicon (Si), manganese (Mn), and aluminium (Al)—require massive amounts of solid carbon not for heat, but as a chemical reductant to strip oxygen away from raw ores.
Currently, these industries rely on fossil-fuel-derived coal, petroleum coke, and coal tar pitch. The obvious green alternative is “biocarbon“—a highly processed, renewable charcoal made from plant biomass. However, creating a drop-in replacement is incredibly difficult. Standard biochar produced from wood is generally too porous, structurally weak, and highly reactive with carbon dioxide (CO2), meaning it burns up or crumbles before it can do its job in a smelting furnace.
To meet the rigorous demands of Si, Mn, and Al production, scientists must re-engineer the entire biochar production process from the ground up. Three recent breakthrough studies reveal how altering reactor designs, exploiting the hidden chemistry of different tree species, and utilizing simple water-washing pretreatments can transform forestry residues into high-performance, metallurgical-grade biocarbon.
Here is a deep dive into the science of making a better biocarbon, based on results from the knowledge-building project BioCarbUpgrade.
1. The Engineering Solution: Steering vapours to trap carbon
When biomass is heated in an oxygen-free environment—a process known as pyrolysis—it thermally decomposes into three distinct phases: solid biochar, condensable liquid bio-oil, and non-condensable gases. If the goal is to produce biocarbon for metallurgical reductants, maximizing the yield of the solid carbon phase is critical.
The dilemma is that achieving the high fixed-carbon content required for industrial applications demands high treatment temperatures, which typically causes more of the biomass mass to escape as volatile gases and liquids, leaving behind very low yields of solid biochar.
A recent study by Kilic et al. (2026) demonstrated that we don’t just have to accept this trade-off. By physically manipulating how hot gases flow inside the pyrolysis reactor, we can force the chemistry to yield significantly more solid carbon.
The Auger Reactor and Counterflow Magic
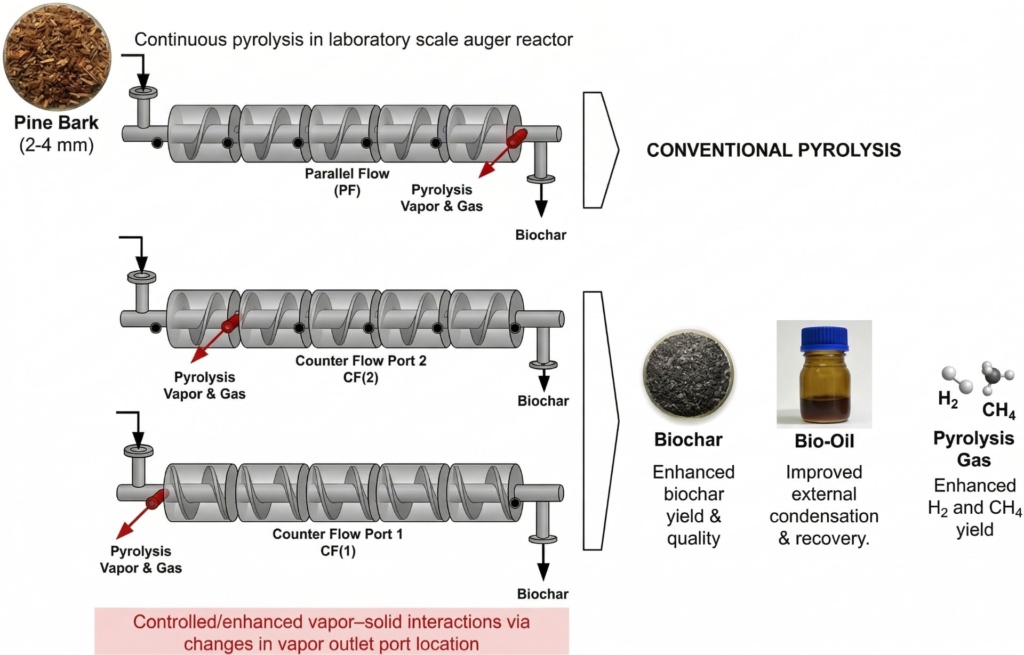
The researchers utilized a continuous laboratory-scale “auger reactor” (essentially a heated screw conveyor) to process pine bark milled to 2-4 millimeter particles. The reactor featured five independently controlled heating zones, allowing the team to test temperatures up to 800°C.
Crucially, the reactor was designed with six different outlet ports, allowing the researchers to change the direction that the evolving hot vapours travelled and their extraction point.
They tested two main vapours configurations against a standard parallel flow baseline:
The conventional method. The raw biomass and the evolving hot vapours travel in the same direction, exiting together at the hottest downstream end of the reactor.
- The vapours are forced to travel backward. As the biomass moves forward into hotter zones, the hot vapours are pushed back toward the entrance, passing directly through the coldest zone over the freshly incoming, unheated pine bark.
- Counterflow 2 (CF2): The vapours are forced to travel backward but was extracted before reaching the coldest zone.
The vapours are forced to travel backward but was extracted before reaching the coldest zone.
The results: Condensation and secondary char
The CF1 configuration yielded spectacular results. By forcing the heavy, reactive, hot organic vapours to pass over the cold incoming pine bark (which sat at temperatures below 200°C), the heavy oils physically condensed directly onto the surface of the raw biomass.
As this oil-coated biomass was conveyed forward into the hotter zones of the reactor, those trapped oils didn’t just re-evaporate; they underwent “secondary reactions”. The heat caused the condensed organic molecules to thermally crack and polymerize into what is known as “secondary char”.
- Yield Increase: The CF1 configuration resulted in a consistent and substantial relative increase in total biochar yield of up to 15.5% compared to the standard PF setup.
- Quality Metrics: This wasn’t just low-quality ash. The increase in biochar yield was accompanied by an increase in fixed carbon yield, meaning the newly formed secondary char was organically enriched.
- Chemical Proof: The researchers also noted a distinct increase in the production of hydrogen (H2) and methane (CH4) gases in the CF configurations. High yields of these specific non-condensable gases are strong indicators of intense secondary tar cracking and char carbonization occurring within the reactor.
The CF2 configuration on the other hand favoured external condensation and achieved the highest bio-oil recovery but also this configuration increased the biochar yield compared to parallel flow.
By simply altering the vapor trajectories, the researchers proved that in situ vapor recirculation is a highly scalable, physical approach to chemically steering pyrolysis pathways toward greater solid carbon recovery.
2. The Chemical Solution: Birch wood’s built-in glue
Even if you optimize the reactor to produce more biochar, you still face a mechanical problem. Normal biochar retains the cellular, microscopic structure of the original plant tissue, making it porous and brittle.
In the metallurgical world, this is unacceptable. For context, carbon anodes used in aluminium (Al) smelting require a compressive strength of 20 to 40 MPa. Fossil-based coke reductants used in the silicon (Si) and manganese (Mn) industries require tensile strengths between 4 and 8 MPa.
To bridge this gap, a study by Johnson et al. (2026) investigated a process to create “Transient Plastic Phase Biochar” (TPPB) from three different woods: birch, spruce, and eucalyptus.
Pressurized Pyrolysis and Pelletization
Instead of heating the wood at atmospheric pressure, the researchers placed wet wood particles (with 75 wt% water added) into sealed reactors. They pressurized the vessels with nitrogen gas to 12.4 MPa and heated them to 320°C for 30 minutes.
This high-pressure environment prevents the structural components of the wood from simply vaporizing. Instead, the biomass undergoes a molten, plastic-like intermediate phase, breaking down the macroscopic cellular morphology of the wood.
The resulting TPPB was then cryo-milled into a fine powder, pressed into dense pellets under 168 MPa of force, and finally calcined (heated) slowly to 900°C.
The Results: The incredible strength of birch
The mechanical testing of these calcined biocarbon pellets revealed astonishing differences based purely on the species of the starting wood:
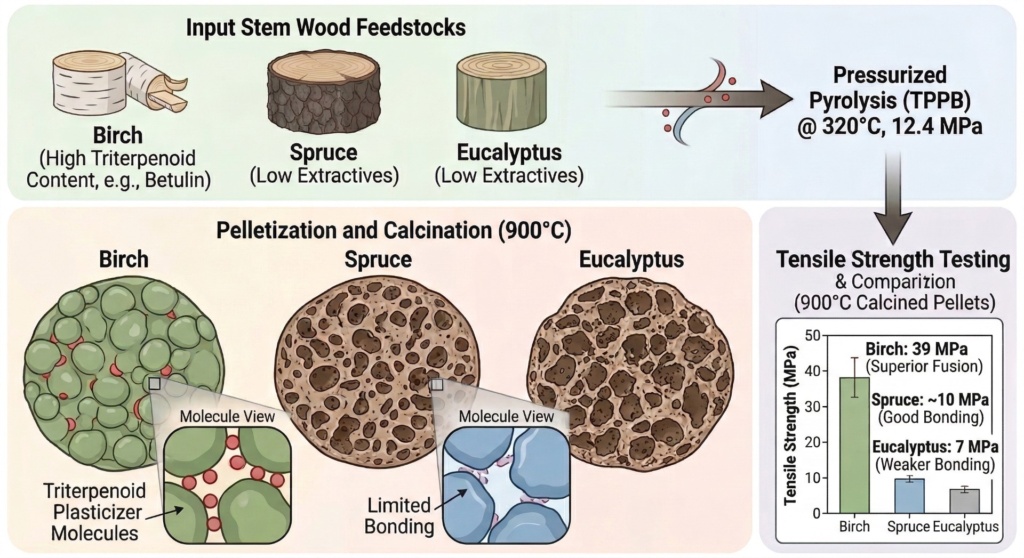
- Eucalyptus biocarbon achieved a tensile strength of 6.86 ± 2.3 MPa.
- Spruce biocarbon achieved a tensile strength of 9.89 ± 2.35 MPa.
- Birch biocarbon completely shattered expectations, achieving a massive tensile strength of 39 ± 4.05 MPa.
To understand why the birch pellets were up to four times stronger than the others, the team utilized advanced techniques including Gas Chromatography-Mass Spectrometry (GC-MS) and solid-state Carbon-13 Nuclear Magnetic Resonance (13C NMR).
They discovered that birch wood contains high concentrations of natural chemical extractives known as triterpenoids, specifically compounds like betulin. These triterpenoid-derived products accounted for over 30% of the extractable material in the birch TPPB, corresponding to roughly 9 wt% of the solid mass. Neither spruce nor eucalyptus contained these compounds.
During the pelletization and calcination phases, these triterpenoid compounds acted as powerful natural plasticizing agents. They effectively lowered the glass transition temperature of the biochar, transforming the powder into a “rubbery phase” at lower temperatures. This allowed the microscopic particles to fuse together seamlessly, generating incredibly strong interparticle bonds without the need for any artificial, external chemical binders.
Furthermore, because the birch particles fused so completely, the resulting biocarbon was significantly less reactive to CO2 (a vital trait so it doesn’t prematurely burn up in a smelting furnace) and exhibited the highest electrical conductivity (5.59 x 103 S/m), which is essential for use as electrodes in aluminium and silicon processing.
3. The Pretreatment Solution: Washing bark and catching liquids
While stem wood like birch is clearly an excellent feedstock, relying exclusively on premium wood to supply massive global metallurgical industries is neither economically nor environmentally sustainable. Alternative, low-grade biomass is desperately needed.
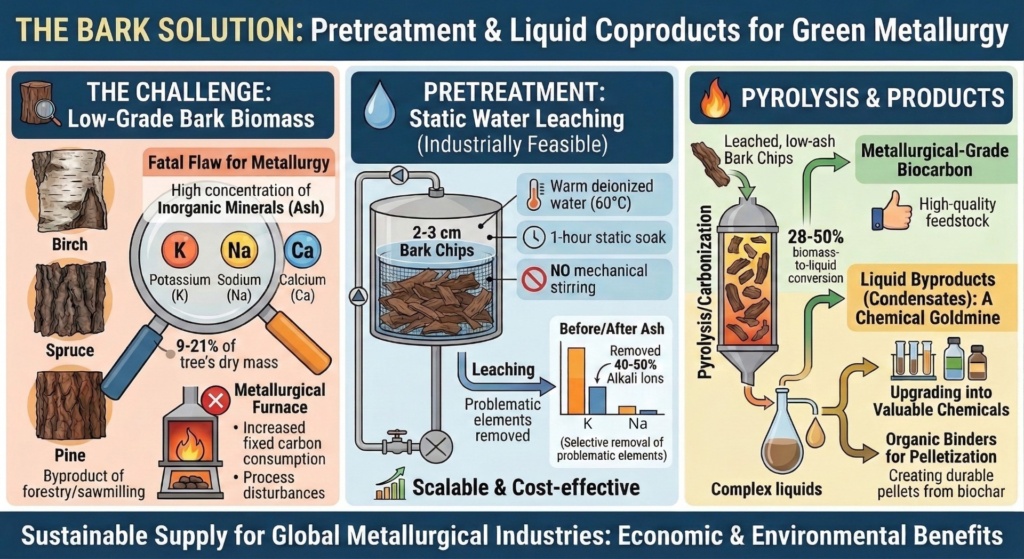
Bark is a prime candidate. It makes up 9 to 21% of a tree’s dry mass and is generated in large amounts as a byproduct of the forestry and sawmilling sectors. However, bark comes with a fatal flaw for metallurgy: it is packed with high concentrations of inorganic minerals (ash), particularly alkaline and alkaline earth metals (AAEMs) like potassium, sodium, and calcium. If introduced into a metallurgical furnace, these impurities can lead to increased fixed carbon consumption, disturb the metallurgical process and influence on the final metal product.
A study by Babinszki et al. (2026) explored how to clean this bark prior to carbonization, and simultaneously analysed the complex liquid byproducts (condensates) generated during its pyrolysis.
Static water leaching: A feasible industrial fix
To remove the problematic minerals, the researchers employed a static water leaching method. They immersed 2 to 3 centimeter chips of birch, spruce, and pine bark in warm deionized water (60°C) for just one hour, without any mechanical stirring.
While grinding the bark into a fine powder would have removed more ash, the researchers specifically chose to use larger chips and a static soak because it represents an industrially feasible, highly scalable, and cost-effective methodology for large operations.
Despite its simplicity, this gentle leaching process was highly effective at targeting the most problematic elements. It successfully removed 40 to 50% of the highly reactive alkali ions (potassium and sodium), alongside a roughly 10% reduction in total ash content.
The liquid byproducts: A chemical goldmine
In the production of biocarbon, roughly 28 to 50% of the biomass mass is converted into condensable liquid bio-oil during pyrolysis. Understanding the chemical makeup of these liquids is vital, as they can be upgraded into valuable chemicals, or even re-used as organic binders for pelletizing weaker biochars.
Using GC/MS analysis, the team compared the condensates from the leached and unleached barks against condensates produced from premium stem wood.
They found clear chemical distinctions:
The total volatile organic content measured in the bark condensates was significantly lower than that of stem wood, yielding far fewer anhydrosugars and carbohydrate decomposition products. This directly correlates with bark having naturally lower cellulose and hemicellulose concentrations than stem wood.
The condensate produced from birch bark contained massively higher concentrations of carboxylic acids—specifically acetic acid—than the condensates from spruce and pine bark. This is driven by the specific molecular structure of broadleaf trees; the main hemicellulose in birch contains numerous O-acetyl side-groups, which cleave off during heating to form high volumes of acetic acid. The researchers also found specific thermal decomposition products like betuligenol and raspberry ketone uniquely in the birch condensates.
Interestingly, the act of leaching the bark slightly altered how it broke down during pyrolysis. By removing the alkali metals (which act as natural catalysts during thermal breakdown), the chemical pathways shifted. The condensates from the leached barks showed increased yields of specific cyclic carbohydrate derivatives, such as furfural, while slightly increasing the yields of lignin-derived phenolic compounds.
Conclusion: The path forward for green metallurgy
The transition away from fossil-fuel reductants in the silicon, manganese, and aluminium industries will not be solved by a single magic bullet. It requires a highly integrated, multidisciplinary approach.
As these three studies demonstrate, achieving metallurgical-grade biocarbon requires us to merge clever reactor engineering (like counterflow vapor recirculation to maximize carbon yield), with a deep understanding of plant chemistry (leveraging the natural triterpenoid plasticizers found in birch wood for structural strength), and practical pretreatments (like static water leaching to strip away problematic ash from cheap, abundant bark).











Comments
No comments yet. Be the first to comment!